Approval
You may have seen stories in the news about people who want access to a drug but can’t get it because that drug has not yet been approved to be used in Canada. It can take 1-2 years for new drugs to be available on the Canadian market after clinical trials conclude.
There are several reasons for this:
- the pharmaceutical company might still be finalizing details of the product’s production, safety protocol, distribution, etc.;
- the pharmaceutical company has to apply to each country to have its product approved for distribution in that country; it must prove with information from the clinical trials and other testing that the drug is of sufficient quality and meets regulatory standards; that it is safe to use, and that it is effective for the disease or condition it is targeting; these applications are very complex and take time to create;
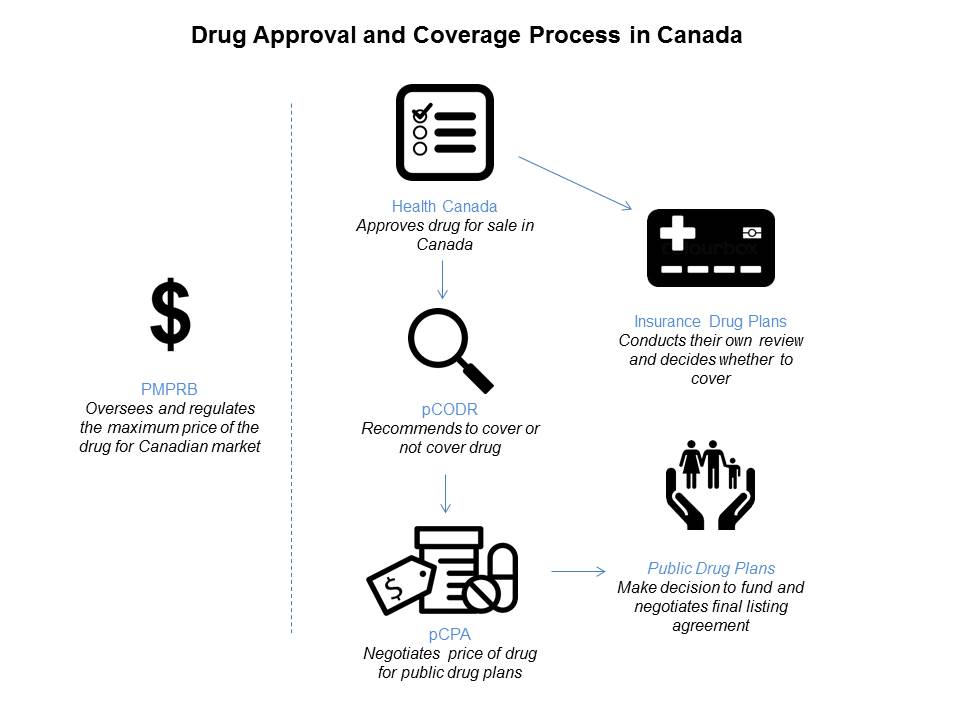
- Health Canada is responsible for reviewing each application for a new drug to be sold in the country; the review process involves many steps and takes time; there is also a high volume of new applications to review; on average, it take 300 business days to complete a review (Source: Health Canada Submission Standards reporting);
- most pharmaceutical companies apply to larger developed nations (e.g. United States, Europe) to market their product before applying to Canada; the reasons vary but usually involve market demands; Canada only represents 2% of the global market for pharmaceutical products (Source: Government of Canada Pharmaceutical Industry profile);
- a drug also needs to be assigned a price by the pharmaceutical company; a federal agency called the Patented Medicines Prices Review Board (PMPRB) then has to determine if the price is reasonable, given the price of the drug on competing international markets and the price of comparable products in Canada (if any); if the asking price is determined to be too high, the pharmaceutical company may decide to appeal; for more information on the PMPRB process, including newly proposed amendments, visit the PMPRB website.
Once Health Canada approves a drug for the Canadian market, it assigns the drug a Drug Identification Number or DIN. This is the unique identifier that is attached to each formulation or dose of the drug. For example, a 100 mg version of the drug will have one number, and the 250 mg version will have another. Health Canada also releases the drug’s Product Monograph, which verifies the intended use for the drug You may be asked for this information when inquiring with your drug plan about coverage of a drug.
To find out if a drug has been approved for sale in Canada, and to access the drug’s DIN and product monograph, you can search for its listing in Health Canada’s Drug Product Database. The database is updated every 24 hours.
We have also provided a list of commonly prescribed cancer treatment drugs and their DIN numbers. Click here to access the list.
If a drug is not approved for sale in Canada, or if a pharmaceutical company decides NOT to attempt to market the drug in Canada, your oncologist may try to access it from outside the country. When a physician requests to bring a drug in to Canada directly, that physician must first receive permission from Health Canada’s Special Access Programme.
Coverage of New Drugs
When a drug is approved for sale in Canada, it does not mean it is automatically paid for by drug plans or hospitals. Each funder conducts a careful evaluation of whether a drug should be funded and whether they have the funds within their budget to pay for it. Each funder also negotiates a final price for a product with the pharmaceutical company, in the form of rebates or other incentives.
Insurance companies use their own internal processes for review and pricing. This means one insurance company might start covering the price of a drug before another company. They all have different processes and timelines.
The provinces, who are responsible for the funding of drugs administered in hospital AND provincial drug plans, look to two pan-Canadian agencies to help them with evaluation and pricing.
This group is part of a broader national non-profit organization called CADTH (Canadian Agency for Drugs and Technologies in Health). pCODR conducts evaluations of new oncology treatment drugs and makes general recommendations on whether provinces should fund each drug. They include feedback from patient non-profit organizations, clinicians, and provincial cancer agencies. Sometimes, pCODR makes a recommendation to fund a drug, contingent on the final price of the drug being lowered.
It takes time to complete these evaluations, and the provinces tend to wait until an evaluation is completed before deciding to cover a drug. This is one of the reasons the provincial drug plans in the Atlantic Provinces tend to add new “take at home” cancer treatment drugs later than insurance drug plans. The average time to complete a review through pCODR is 8 months.
pCODR provides a list of all the drugs they are reviewing or have reviewed. You can find it here.
The pCPA conducts negotiations on the final price of new brand name drug on behalf of the provinces. This includes new oncology treatment drugs. The time it takes to conduct negotiations varies by drug and can last anywhere from 2-12 months.
Once negotiations are complete, each province still needs to decide if it has the BUDGET to cover the cost of the drug on its hospital list, or its provincial drug plan list. This can also take time to process. A province holds the right NOT to cover a drug if it feels it cannot afford it or it disagrees with the pCODR and pCPA decisions.